ASCO Report of Pioneering Treatment of Lymphopenia with Significant Overall Survival Benefit in Advanced Pancreatic Cancer
- While Epogen and Neupogen addresses anemia and neutropenia, no therapy has existed for the treatment of lymphopenia until ANKTIVA® –The Cancer BioShield.
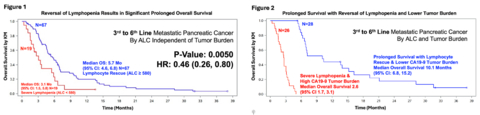
- Landmark overall survival benefit (P-value 0.005, HR: 0.46 [0.26, 0.80]) observed in 3rd-6th line metastatic pancreatic cancer, correlated with reversal of lymphopenia.
- Patients with lower tumor burden and improved lymphocyte counts (ALC ≥1,000) had a median overall survival of 10.1 months, supporting earlier intervention in the disease course.
ImmunityBio, Inc. ((IBRX) today announced results presented at ASCO 2025 of the first known treatment for lymphopenia with ANKTIVA and CAR-NK therapy. This data supports that reversal of lymphopenia, a well-established root cause of early mortality in patients with cancer across all tumor types, correlates with significant improved survival.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250603288322/en/
While anemia and neutropenia have long been addressed by agents like Epogen and Neupogen, no therapy has ever existed for lymphopenia—until now. ANKTIVA (nogapendekin alfa inbakicept-pmln), an IL-15 superagonist approved in April 2024 for BCG-unresponsive non-muscle- invasive bladder cancer carcinoma in situ with or without papillary disease, represents the first lymphocyte-stimulating agent (LSA) capable of expanding lymphocytes critical for immunogenic cell death, such as natural killer (NK) and T cells.
These findings emphasize the need for a therapy to overcome treatment induced lymphopenia with higher mortality as presented at ASCO 2025 by several institutions (Abstract #8054, Satoskar et al. and Abstract #2663, Saleh et al.)
In a single-arm QUILT-88 clinical trial of 86 participants with third-to-sixth-line metastatic pancreatic cancer with very high tumor burden (CA19-9 levels exceeding 34,000 IU/ml), for which no therapy currently exists, patients received ANKTIVA subcutaneously in combination with off-the-shelf, ex-vivo infusion of CAR-NK cells (PD-L1 t-haNK) and low dose immuno-modulating chemotherapy. This first reported study of treating lymphopenia demonstrated significant differences in median overall survival in subjects whose lymphopenia was reversed (Absolute Lymphocyte Count: ALC ≥ 1,000).
In 67 out of 86 subjects, reversal of lymphopenia was achieved and median overall survival was significantly prolonged compared to those who remained in severe lymphopenia with P-value 0.005, HR: 0.46 (0.26, 0.80) in Figure 1. In subjects with lymphopenia rescue and a lower tumor burden (less than the median CA19-9 of 34,000 IU/mL), the median overall survival in these very advanced metastatic pancreatic cancer patients exceeded 10 months (Figure 2).
These findings of improved survival in pancreatic cancer patients with lower tumor burden point to the potential of further prolonged overall survival in pancreatic cancer patients if treated in the first line or neoadjuvant stages of disease. Highlighting the importance of lymphopenia reversal, Oncologist published a peer-reviewed paper titled "Recurrent pancreatic cancer treated with N-803 and PD-L1 t-haNK followed by an EGFR-targeted nanocell drug conjugate," demonstrating that in a patient with 2nd line metastatic pancreatic cancer treated with the full Cancer BioShield platform—including ANKTIVA, CAR-NK cells (PD-L1 t-haNK), and antigen-targeting adenoviruses—remained in remission for over six years and maintains a high quality of life at the date of this release.
The expanded access authorization announced yesterday enables patients across all solid tumor types who have exhausted first-line therapy including chemotherapy, radiation, or immunotherapy to receive Anktiva as a lymphocyte stimulating agent to protect the immune system from the lymphogenic adverse effects of current standards of care.
The ASCO Annual Meeting 2025 materials from ImmunityBio can be found below:
Association of lymphopenia rescue and CA19-9 levels with overall survival following IL-15 superagonist N-803 and PD-L1 t-haNK chemo-immunotherapy for 3rd line or greater metastatic pancreatic cancer.
Abstract Text: https://meetings.asco.org/abstracts-presentations/246953
Poster PDF: https://immunitybio.com/asco-2025-a/
About the Cancer BioShield™ Platform
The Cancer BioShield platform is a first-in-class immunotherapy strategy designed to restore immune competence by reversing lymphopenia—the loss of functional immune cells caused by cancer itself and by conventional treatments such as chemotherapy, radiation and immunotherapy. At its core is ANKTIVA® (nogapendekin alfa inbakicept-pmln), an IL-15 agonist approved for BCG-unresponsive non-muscle-invasive bladder cancer CIS with or without papillary disease, activates and proliferates natural killer (NK) cells and CD4+ and CD8+ T cells, restoring lymphocyte levels critical for immunosurveillance, immunogenic cell death, and long-term tumor control.
The platform employs a multi-modal approach:
- In-vivo stimulation: Subcutaneous administration of ANKTIVA expands NK and T cells, boosting anti-tumor immunity.
- Ex-vivo targeted cytotoxicity: Off-the-shelf PD-L1 t-haNK CAR-NK cells are engineered to target and eliminate PD-L1–expressing tumor cells and immunosuppressive neutrophils (myeloid-derived suppressor cells), enhancing anti-tumor specificity and reducing immune evasion.
- Memory Cytokine-Enriched Natural Killer (M-ceNK) cell therapy: M-ceNK cells are developed via cytokine activation and expansion of autologous and allogeneic NK cells collected through apheresis, potentially providing long-term immune memory and sustained cytotoxic capacity.
Together, these components offer a comprehensive, novel, immune-restoring therapeutic platform aimed at not only expanding effector immune cells but also overcoming tumor-mediated immune suppression to support long-term disease control.
The platform's effectiveness can be tracked through universally utilized, simple complete blood count (CBC): increases in absolute lymphocyte count (ALC) reflect ANKTIVA's lymphocyte-stimulating activity, while reductions in the neutrophil-to-lymphocyte ratio (NLR) demonstrate PD-L1 t-haNK's immunosuppressive neutrophil targeting. Low ALC and high NLR levels are laboratory measurements that have been extensively reported as predictive biomarkers of poor prognosis with early mortality across all tumor types5,6. The data presented by ImmunityBio for the first time demonstrates that improving ALC and NLR correlates with significant enhanced overall survival and clinical benefit.
About Lymphopenia and Absolute Lymphocyte Count (ALC)
Lymphopenia—the loss of key immune cells such as NK, CD4+, and CD8+ T cells—is a common side effect of chemotherapy1, radiation2,3, and some immunotherapies4. Unlike anemia and neutropenia, which have FDA-approved treatments like EPO and Neupogen, no therapy previously existed to treat this immune cell depletion. Lymphopenia weakens the immune system, increases infection risk, and is linked to early death across many cancer types5. Low Absolute Lymphocyte Count (ALC) is a recognized poor prognostic marker. ANKTIVA® is the first approved therapy to restore lymphocyte levels by activating and expanding NK and T cells—without increasing immunosuppressive T regulatory cells7.
More information on lymphopenia could be found on Twitter/X @DrPatSoonShiong articles here: https://x.com/DrPatSoonShiong/articles
References:
- Ray-Coquard I, et al. Lymphopenia as a prognostic factor for overall survival in advanced carcinomas, sarcomas, and lymphomas. Cancer Res. 2009 Jul 1;69(13):5383-91. doi: 10.1158/0008-5472.CAN-08-3845. Epub 2009 Jun 23. PMID: 19549917; PMCID: PMC2775079.
- Chen D, et al. Absolute Lymphocyte Count Predicts Abscopal Responses and Outcomes in Patients Receiving Combined Immunotherapy and Radiation Therapy: Analysis of 3 Phase 1/2 Trials. Int J Radiat Oncol Biol Phys. 2020 Sep 1;108(1):196-203. doi: 10.1016/j.ijrobp.2020.01.032. Epub 2020 Feb 7.
- Pike LRG, et al. The Impact of Radiation Therapy on Lymphocyte Count and Survival in Metastatic Cancer Patients Receiving PD-1 Immune Checkpoint Inhibitors. Int J Radiat Oncol Biol Phys. 2019 Jan 1;103(1):142-151. doi: 10.1016/j.ijrobp.2018.09.010. Epub 2018 Sep 15. PMID: 30227198.
- Lee, Y.J., et al. Peripheral lymphocyte count as a surrogate marker of immune checkpoint inhibitor therapy outcomes in patients with non-small-cell lung cancer. Sci Rep 12, 626 (2022). https://doi.org/10.1038/s41598-021-04630-9
- Ménétrier-Caux C., et al. Lymphopenia in Cancer Patients and its Effects on Response to Immunotherapy: an opportunity for combination with Cytokines? J Immunother Cancer. 2019 Mar 28;7(1):85. doi: 10.1186/s40425-019-0549-5. PMID: 30922400; PMCID: PMC6437964.
- Templeton AJ, et al. Prognostic role of neutrophil-to-lymphocyte (NLR) ratio in solid tumors: a systematic review and meta-analysis. J Natl Cancer Inst. 2014 May 29;106(6):dju124. doi: 10.1093/jnci/dju124. PMID: 24875653.
- FDA ANKTIVA Label, April 2024 - https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/761336s000lbl.pdf
About ImmunityBio
ImmunityBio is a vertically-integrated biotechnology company developing next-generation therapies and vaccines that bolster the natural immune system to defeat cancers and infectious diseases. The Company's range of immunotherapy and cell therapy platforms, alone and together, act to drive and sustain an immune response with the goal of creating durable and safe protection against disease. Designated an FDA Breakthrough Therapy, ANKTIVA is the first FDA-approved immunotherapy for non-muscle invasive bladder cancer CIS that activates natural killer cells, T cells, and memory T cells for a long-duration response. The Company is applying its science and platforms to treating cancers, including the development of potential cancer vaccines, as well as developing immunotherapies and cell therapies that we believe sharply reduce or eliminate the need for standard high-dose chemotherapy. These platforms and their associated product candidates are designed to be more effective, accessible, and easily administered than current standards of care in oncology and infectious diseases. For more information, visit ImmunityBio.com (Founder's Vision) and connect with us on X (Twitter), Facebook, LinkedIn, and Instagram.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, such as statements regarding clinical trial data and potential results and implications to be drawn therefrom, the expectation that the EAP described herein will enable access to ANKTIVA for patients across all solid tumor types who have exhausted first-line therapy including chemo, radiation or immunotherapy, the RMAT designation as previously reported and potential results therefrom and regulatory submissions in connection therewith, the belief that ALC levels and NLR levels obtained from a CBC are predictors of clinical benefit and outcomes relating to overall survival, the belief that improving ALC levels and NLR levels correlates with enhanced overall survival and clinical benefit, the belief that reversal of lymphopenia correlates with improved survival, clinical trial and expanded access program enrollment, data and potential results to be drawn therefrom, anticipated components of ImmunityBio's Cancer BioShield platform, the development of therapeutics for cancer and infectious diseases, potential benefits to patients, potential treatment outcomes for patients, the described mechanism of action and results and contributions therefrom, potential future uses and applications of ANKTIVA alone or in combination with other therapeutic agents for the prevention or reversal of lymphopenia, potential future uses and applications of ANKTIVA alone or in combination with other therapeutic agents across multiple tumor types and indications and for potential applications beyond oncology, potential regulatory pathways and the regulatory review process and timing thereof, the application of the Company's science and platforms to treat cancers or develop cancer vaccines, immunotherapies and cell therapies that has the potential to change the paradigm in cancer care, and ImmunityBio's approved product and investigational agents as compared to existing treatment options, among others. Statements in this press release that are not statements of historical fact are considered forward-looking statements, which are usually identified by the use of words such as "anticipates," "believes," "continues," "goal," "could," "estimates," "scheduled," "expects," "intends," "may," "plans," "potential," "predicts," "indicate," "projects," "is," "seeks," "should," "will," "strategy," and variations of such words or similar expressions.
Statements of past performance, efforts, or results of our preclinical and clinical trials, about which inferences or assumptions may be made, can also be forward-looking statements and are not indicative of future performance or results. Forward-looking statements are neither forecasts, promises nor guarantees, and are based on the current beliefs of ImmunityBio's management as well as assumptions made by and information currently available to ImmunityBio. Such information may be limited or incomplete, and ImmunityBio's statements should not be read to indicate that it has conducted a thorough inquiry into, or review of, all potentially available relevant information. Such statements reflect the current views of ImmunityBio with respect to future events and are subject to known and unknown risks, including business, regulatory, economic and competitive risks, uncertainties, contingencies and assumptions about ImmunityBio, including, without limitation, (i) risks and uncertainties regarding the FDA regulatory submission, filing and review process and the timing thereof, (ii) whether the RMAT designation will lead to an accelerated review or approval, of which there can be no assurance, (iii) risks and uncertainties regarding commercial launch execution, success and timing, (iv) risks and uncertainties regarding participation and enrollment and potential results from the expanded access clinical investigation program described herein, (v) whether clinical trials will result in registrational pathways and the risks, (vi) whether clinical trial data will be accepted by regulatory agencies, (vii) the ability of ImmunityBio to continue its planned preclinical and clinical development of its development programs through itself and/or its investigators, and the timing and success of any such continued preclinical and clinical development, patient enrollment and planned regulatory submissions, (viii) potential delays in product availability and regulatory approvals, (ix) ImmunityBio's ability to retain and hire key personnel, (x) ImmunityBio's ability to obtain additional financing to fund its operations and complete the development and commercialization of its various product candidates, (xi) potential product shortages or manufacturing disruptions that may impact the availability and timing of product, (xii) ImmunityBio's ability to successfully commercialize its approved product and product candidates, (xiii) ImmunityBio's ability to scale its manufacturing and commercial supply operations for its approved product and future approved products, and (xiv) ImmunityBio's ability to obtain, maintain, protect, and enforce patent protection and other proprietary rights for its product candidates and technologies. More details about these and other risks that may impact ImmunityBio's business are described under the heading "Risk Factors" in the Company's Form 10-K filed with the U.S. Securities and Exchange Commission (SEC) on March 3, 2025, and the Company's Form 10-Q filed with the SEC on May 12, 2025, and in subsequent filings made by ImmunityBio with the SEC, which are available on the SEC's website at www.sec.gov. ImmunityBio cautions you not to place undue reliance on any forward-looking statements, which speak only as of the date hereof.
View source version on businesswire.com: https://www.businesswire.com/news/home/20250603288322/en/
ImmunityBio Contacts:
Investors
Hemanth Ramaprakash, PhD, MBA
ImmunityBio, Inc.
+1 858-746-9289
[email protected]
Media
Sarah Singleton
ImmunityBio, Inc.
+1 415-290-8045
[email protected]
